- 著者
- Munenobu Kashiwa Ryo Matsushita
- 出版者
- The Pharmaceutical Society of Japan
- 雑誌
- Biological and Pharmaceutical Bulletin (ISSN:09186158)
- 巻号頁・発行日
- vol.45, no.7, pp.895-903, 2022-07-01 (Released:2022-07-01)
- 参考文献数
- 28
- 被引用文献数
- 2
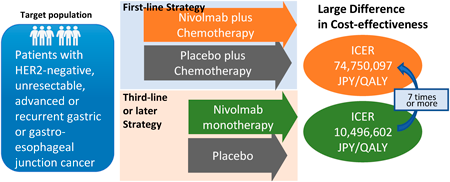
A model-based, cost-effectiveness analysis was conducted to evaluate the difference in cost-effectiveness of nivolumab (NIVO), between first-line therapy in combination with chemotherapy and third-line or later monotherapy for patients with unresectable, advanced or recurrent gastric or gastro-esophageal junction cancer, with the aim of supporting the economic evaluation of healthcare in Japan. Data on overall survival and progression-free survival were obtained from the phase 3 clinical trials, ATTRACTION-4 and ATTRACTION-2. A partitioned survival model was developed to predict costs and outcomes. Direct medical costs were considered from the perspective of the Japanese national health insurance (NHI) payer. The model time horizon was set to 10 years. Health outcomes were defined as life years (LYs) and quality-adjusted life years (QALYs) gained. The incremental cost-effectiveness ratio (ICER) of NIVO compared to the control group was estimated. Sensitivity analyses were performed to assess the uncertainty of parameter setting. A willingness-to-pay threshold of 15 million JPY (Japanese yen) was used. Compared to each control group, the ICERs for NIVO treatment per 1 LY gained were 65745714 JPY for first-line treatment, 7420202 JPY for third-line or later treatment, and 74750097 JPY and 10496602 JPY per QALY gained, respectively. Probabilistic sensitivity analyses estimated that the probability of NIVO treatment being cost-effective for first-line and third-line treatment was 23.5 and 74.3%, respectively. From the perspective of the Japanese NHI payer, NIVO was cost-effective as third-line or later monotherapy for patients with advanced gastric cancer, but not in combination with first-line chemotherapy.