- 著者
- Dang Huy HIEP Yuta TANAKA Hiroki MATSUBARA Shoji ISHIZAKA
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.10, pp.1275-1278, 2020-10-10 (Released:2020-10-10)
- 参考文献数
- 18
- 被引用文献数
- 4
This paper describes a novel method for fabricating paper-based microfluidic devices using a laser beam scanning technique. Cellulose chromatography papers were treated with octadecyltrichlorosilane (OTS) to make them entirely hydrophobic. A photoacid generator (CPI-410S) was soaked into the paper, and irradiated with a 405-nm laser beam to induce acid generating reactions. Since the silyl ether bond between cellulose and OTS was cleaved by the hydrolysis reaction, the photo-irradiated area changed to hydrophilic. By scanning the laser beam using a Galvo mirror system, arbitrary shaped hydrophilic patterns were successfully created on the paper in 50 μm resolution. To the best of our knowledge, this is the first report on the fabrication of hydrophilic channels on the OTS-treated paper using photo-induced acid generation processes coupled with the laser beam scanning technique. Quantification of nitrite was demonstrated with the paper device made by this method.
- 著者
- Akihiro JO Sae KOYAGI Wataru HOBO Saori OTANI Shoujiro OGAWA Tatsuya HIGASHI
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.9, pp.1099-1104, 2020-09-10 (Released:2020-09-10)
- 参考文献数
- 15
- 被引用文献数
- 13
Liquid chromatography/electrospray ionization–tandem mass spectrometry (LC/ESI-MS/MS) enables the accurate and precise quantification of various analytes at very low concentrations, but it has room for improvement in analysis throughput. Multiplexing of samples in the same injection could be a promising procedure for enhancing the analysis throughput. This could be achieved by derivatization of the multiple samples with multiple isotopologous reagents. In this study, a sample-multiplexed LC/ESI-MS/MS assay using the 1-[(4-dimethylaminophenyl)carbonyl]piperazine (DAPPZ) isotopologues (2H0-, 2H3-, and 2H6-forms) was developed and validated for the simultaneous determination of primary bile acids in three different plasma samples in a single run. The developed method had satisfactory intra- and inter-assay precisions (≤ 2.3 and ≤ 4.2%, respectively) and accuracy (99.0 – 100.3%), and could reduce the total LC/ESI-MS/MS run time by more than 60% for 42 samples compared to the conventional method. Thus, the derivatization with the DAPPZ isotopologues worked well for enhancing the throughput of the LC/ESI-MS/MS assay of the bile acids.
- 著者
- Tomoko FUKUUCHI Shun-suke MORIYA Toru SUGIYAMA Hidetsugu TABATA Kiyoko KANEKO
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- pp.20P252, (Released:2020-09-11)
- 被引用文献数
- 1
- 著者
- Adrian ARENDOWSKI Krzysztof OSSOLINSKI Joanna NIZIOL Tomasz RUMAN
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- pp.20P226, (Released:2020-08-21)
- 被引用文献数
- 13
- 著者
- Ze Li Jing Zhang Yunjia Yang Huilan Xu Jianwu Wang Yi Yang
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- pp.20N013, (Released:2020-08-21)
- 被引用文献数
- 5
- 著者
- Yusuke KITAMURA Kotaro MISHIO Pelin ARSLAN Boui IKEDA Chiharu IMOTO Yousuke KATSUDA Toshihiro IHARA
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.8, pp.959-964, 2020-08-10 (Released:2020-08-10)
- 参考文献数
- 12
- 被引用文献数
- 6
Ferrocene (Fc) and β-cyclodextrin (βCyD) were modified at each end of stem-loop structured DNA as an electrochemical signal generator and its quencher, respectively, to give an electrochemical molecular beacon (eMB). A relatively high efficiency of signal quenching was achieved by an inclusion complex (βCyD ⊃ Fc) formation that was induced on the stem structure of the closed form (= stem-loop structure) of eMB. With the addition of target DNA, the structure of eMB opened to form a linear duplex, where the Fc dissociated from the βCyD to restore its intrinsic electrochemical signal. The signal contrast of the electric current for this off/on-type sensor was high, ca. 95. This technique did not require any modification of the electrode surface, and it realized the detection of the target nucleic acids in a homogeneous solution with a high sensitivity using high-performance liquid chromatography (HPLC) equipped with electrochemical detector.
- 著者
- Hiroka SUGAI Shunsuke TOMITA Ryoji KURITA
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.8, pp.923-934, 2020-08-10 (Released:2020-08-10)
- 参考文献数
- 105
- 被引用文献数
- 13
To capture a broader scope of complex biological phenomena, alternatives to conventional sensing based on specificity for cell detection and characterization are needed. Pattern-recognition-based sensing is an analytical method designed to mimic mammalian sensory systems for analyte identification based on the pattern recognition of multivariate data, which are generated using an array of multiple probes that cross-reactively interact with analytes. This sensing approach is significantly different from conventional specific cell sensing based on highly specific probes, including antibodies against biomarkers. Encouraged by the advantages of this technique, such as the simplicity, rapidity, and tunability of the systems without requiring a priori knowledge of biomarkers, numerous sensor arrays have been developed over the past decade and used in a variety of cell sensing applications; these include disease diagnosis, drug discovery, and fundamental research. This review summarizes recent progress in pattern-recognition-based cell sensing, with a particular focus on guidelines for designing materials and arrays, techniques for analyzing response patterns, and applications of sensor systems that are focused primarily for the biomedical field.
- 著者
- Shiori TAKASHINA Yuki IGARASHI Miki TAKAHASHI Yukie KONDO Koichi INOUE
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- pp.20N015, (Released:2020-07-24)
- 被引用文献数
- 10
1 0 0 0 OA Building a Low-cost Standalone Electrochemical Instrument Based on a Credit Card-sized Computer
- 著者
- Toshi NAGATA Kentaro SUZUKI
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.34, no.10, pp.1213-1216, 2018-10-10 (Released:2018-10-10)
- 参考文献数
- 17
- 被引用文献数
- 3
A low-cost, standalone electrochemical instrument was built from a credit card-sized computer and inexpensive A/D and D/A converter chips. The instrument is capable of cyclic voltammetry and constant potential electrolysis, with the potential range of –4 to +4 V and the current range of 1 μA to 20 mA.
- 著者
- Zhuoliang LIU Tianxiong LIU Cheng-an TAO Xianzhe CHEN Jian HUANG Fang WANG Jianfang WANG
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.7, pp.835-840, 2020-07-10 (Released:2020-07-10)
- 参考文献数
- 37
- 被引用文献数
- 3
Sensitive and specific detection of nucleic acids or proteins, which act as biomarkers, is of great importance in disease diagnosis. By combing the concept and operation of an endonuclease-assisted target-responsive amplification method and peroxidase-mimic DNAzyme generated by terminal deoxynucleotidyl transferase (TdT), a novel and facile colorimetric biosensor was developed for DNA and protein. Target DNA and thrombin were chosen as representative biomolecules. The production of cleavage fragments can only be triggered by specific target binding and the following nicking process, which do not occur spontaneously. In the signal collection part, numerous guanine-rich DNA were produced through the prolongation of cleavage fragments by TdT and formed highly effective DNAzyme with hemin. In this novel amplification method, we succeeded in realizing sensitive and specific detection of target DNA and thrombin. Under optimal conditions, target DNA can be detected as low as 1 pM, and thrombin with a detection limit of 100 pM. The method also proves the potential versatility and feasibility of TdT-generated DNAzyme in various bio-analyses.
- 著者
- Piyawan PAISRISARN Takao YASUI Yoshinobu BABA
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.7, pp.791-798, 2020-07-10 (Released:2020-07-10)
- 参考文献数
- 98
- 被引用文献数
- 4
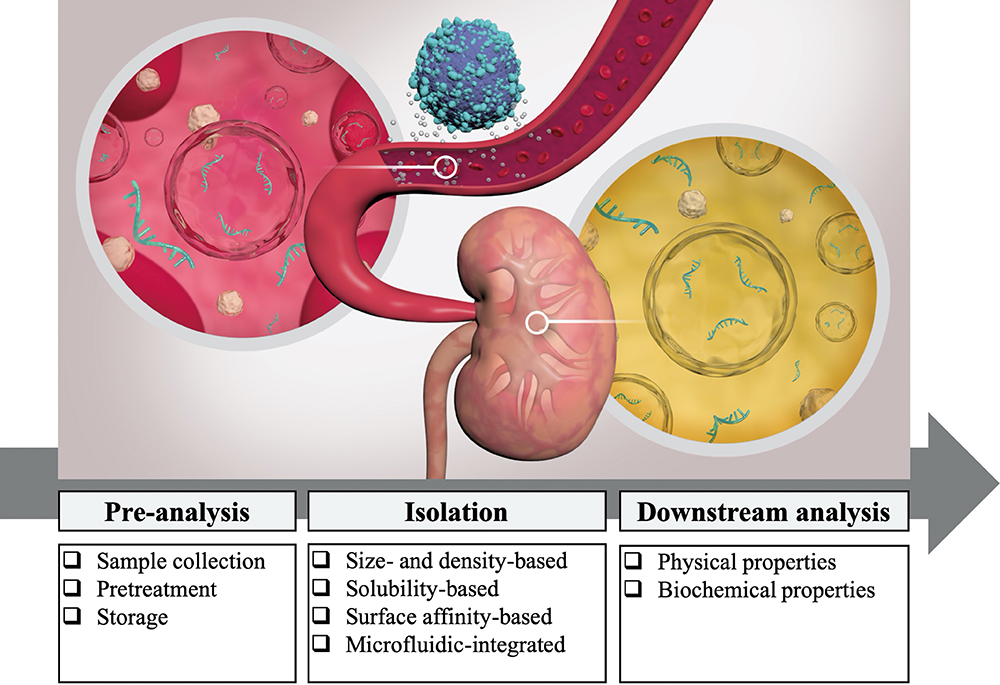
Extracellular vesicles (EVs) play an important role in cell-to-cell communication by carrying molecular messages that reflect physiological and pathological conditions of the parent cells. EVs have been identified in all body fluids; and among them, urine stands out as a sample that is easy and inexpensive to obtain and can be collected over time to monitor changes. Various protocols have been established to study urinary extracellular vesicles (UEVs) and they have shown great potential as a biomarker source for clinical applications, not only for urological, but also non-urological diseases. Due to the high variability and low reproducibility of pre-analytical and analytical methods for UEVs, establishing a standardized protocol remains a challenge in the field of diagnosis. Here, we review UEV studies and present the techniques that are most commonly used, those that have been applied as new developments, and those that have the most potential for future applications. The workflow procedures from the sampling step to the qualitative and quantitative analysis steps are summarized along with advantages and disadvantages of the methodologies, in order to give consideration for choosing the most promising and suitable method to analyze human UEVs.
- 著者
- Shuyi SUN Kyohei MATSUI So TANABE Dung NGUYEN Takamasa KINOSHITA Yojiro YAMAMOTO Hiroshi SHIIGI
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.7, pp.787-789, 2020-07-10 (Released:2020-07-10)
- 参考文献数
- 11
- 被引用文献数
- 4
In this reported work, we achieved high-throughput, highly sensitive fluorescent analysis using an enzyme-linked immunosorbent assay (ELISA) that employed a metallic nanoparticle (NP)-immobilized 96-well plate. The immobilization of metallic NPs on a 96-well plate effectively amplified fluorescent signals of the assay. The silver (Ag) NP-immobilized plate showed the best fluorescent enhancement effect of all plates immobilized by metal NPs. Our results demonstrate the potential of applying Ag NPs to enhance the efficiency of direct and indirect ELISA by the labeling of targets.
- 著者
- Vivian Bee Chin LEE Noor Faizah MOHD-NAIM Eiichi TAMIYA Minhaz Uddin AHMED
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.34, no.1, pp.7-18, 2018-01-10 (Released:2018-01-10)
- 参考文献数
- 85
- 被引用文献数
- 1 76
Electrochemical bio-sensing using paper-based detection systems is the main focus of this review. The different existing designs of 2-dimensional and 3-dimensional sensors, and fabrication techniques are discussed. This review highlights the effect of adopting different sensor designs, distinct fabrication techniques, as well as different modification methods, in order to produce reliable and reproducible reading. The use of various nanomaterials have been demonstrated in order to modify the surface of electrodes during fabrication to further enhance the signal for subsequent analysis. The reviewed sensors were classified into categories based on their applications, such as diagnostics, environmental and food testing. One of the major advantages of using paper-based electrochemical sensors is the potential for miniaturization, which only requires relatively small amount of samples, and the low cost for the purpose of mass production. Additionally, most of the devices reviewed were made to be portable, making them well-suited for on-site detection. Finally, paper-based detection is an ideal platform to fabricate cost-effective, user-friendly and sensitive electrochemical biosensors, with large capacity for customization depending on functional needs.
- 著者
- M. Lutfi FIRDAUS Ikka FITRIANI Santhy WYANTUTI Yeni W. HARTATI Renat KHAYDAROV Jason A. MCALISTER Hajime OBATA Toshitaka GAMO
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.33, no.7, pp.831-837, 2017-07-10 (Released:2017-07-10)
- 参考文献数
- 60
- 被引用文献数
- 2 100
Novel green-chemistry synthesis of silver nanoparticles (AgNPs) is introduced as a low-cost, rapid and easy-to-use analytical method for mercury ion detection. Aqueous fruit extract of water apple (Syzygium aqueum) was used for the first time as bioreductant to synthesize stable AgNPs. The prepared AgNPs have a yellowish-brown color with a surface plasmon resonance peak at 420 nm. The addition of Hg(II) ions then changes the AgNPs color to colorless. The color change was in proportion to the concentration of Hg(II) ions. The presence of other metal ions in the system was also evaluated. The proposed method shows good selectivity and sensitivity towards Hg(II) ions. Using UV-visible spectrophotometry, the detection limit of the developed method was 8.5 × 10−7 M. The proposed method has been successfully applied for determination of Hg(II) ions in tap and lake water samples with precision better than 5%.
- 著者
- Tomoyuki YASUKAWA Asa MORISHIMA Masato SUZUKI Junya YOSHIOKA Keitaro YOSHIMOTO Fumio MIZUTANI
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.35, no.8, pp.895-901, 2019-08-10 (Released:2019-08-10)
- 参考文献数
- 39
- 被引用文献数
- 2 7
We applied a fabrication method for the formation of island organization of cells based on a three-dimensional (3D) device for negative dielectrophoresis (n-DEP) to produce cell aggregates with uniform numbers of cells rapidly and simply. The intersections formed by rotating the interdigitated array (IDA) with two combs of band electrodes on the upper substrate by 90° relative to the IDA with two combs on the lower substrate were prepared in the device. The AC voltage was applied to a comb on the upper substrate and a comb on the lower substrate, while AC voltage with opposite phase was applied to another comb on the upper substrate and another comb on the lower substrate. Cells dispersed randomly were directed toward the intersections with relatively lower electric fields due to n-DEP, which formed by AC voltage applied bands with the identical phase, resulting in the formation of island patterns of cells. The cells accumulated at intersections were promoted to form the cell aggregates due to the close contact together. The production of cell aggregations adhered together was easily found by the dispersion behavior after switching the applied frequency to convert the cellular pattern. When cells were accumulated at the intersections by n-DEP for 45 min, almost accumulations of cells were adhered together, and hence a formations of cell aggregations. By using the present method, we can rapidly and simply fabricate cell aggregations with a uniform number of cells.
- 著者
- Tetsuji YAMAGUCHI Tetsuya MORI Kengo AOKI Ryutaro ODA Masatoshi YASUTAKE Ayako NAKAMURA Kayori TAKAHASHI Tomoo SIGEHUZI Haruhisa KATO
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.6, pp.761-768, 2020-06-10 (Released:2020-06-10)
- 参考文献数
- 15
- 被引用文献数
- 2
This paper presents a study of the size distributions of colloidal nanoparticles using an online dynamic light scattering (DLS) unit with a uni-tau multi-bit correlator (UMC) combined with a centrifugal field-flow fractionation (CF3) separator. Conventionally, the FFF-UV-MALS system utilizing field-flow fractionation (FFF) combined with a UV detector and multi-angle light scattering instrument (MALS) could be used to obtain the particle size distribution of colloidal nanoparticles. Lately, DLS as a technique to measure the size distributions of colloid materials has become prevalent. However, the DLS instrument will practically measure only the large particles in a multi-modal particle mixture. Therefore, the CF3-DLS w/UMC system that was developed consisted of a CF3 unit connected to an online DLS instrument with UMC. The system could measure the volume- or number-based size distribution with highly quantitative and accurate histograms for multi-modal samples. The size distributions were validated with size distributions obtained by images of an atomic force microscope (AFM). Two types of colloidal silica nanoparticles with different distribution widths were used in this study.
- 著者
- Hongying CHENG Siyu XIA Yujie ZHOU Binbin LIN
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.6, pp.745-751, 2020-06-10 (Released:2020-06-10)
- 参考文献数
- 43
We describe here a magnetic molecular imprinted polymeric ionic liquid (MMIPIL) film by using a functionalized ionic liquid (3-vinyl-4-amino-5-imidazole carboxamide chloride, IL) and Fe3O4@Polyrutin–COOH as a functional monomer and supporting materials. The change in the direction of the charge density in the structure of MMIPIL polymer resulted in a red shift of about 100 nm for the characteristic group of –C=O. Polyrutin containing an electron-rich benzene ring and multiple hydroxyl groups not only prevented the aggregation of Fe3O4, but also benefitted to immobilize template molecules. More symmetric amino groups in the template molecules generated more hydrogen bonds and other synergistic effects between MEL and the functional monomers, which resulted in a highly-matched and highly stable MMIPIL sensor. The proposed magnetic sensor lowered the matching potential, and enhanced the signal for the detection of melamine (MEL) in milk powder. Under the optimum conditions, the MEL template molecule showed a significant linear relationship between 5.0 × 10−3 and 0.8 μg/L with a detection limit (S/N = 3) of 1.5 × 10−3 μg/L. The MMIPIL sensor showed wonderful selectivity and exhibited facile, fast and efficient results in the monitoring MEL with recoveries of between 96.5 and 108.3%.
- 著者
- Monu KUMAR Neha GUPTA Amit Pratap SINGH
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.6, pp.659-666, 2020-06-10 (Released:2020-06-10)
- 参考文献数
- 65
- 被引用文献数
- 8
Two novel malonyl-based chemosensors, N,N′-bis(ethyl-4′-benzoate)-1,3-propanediamide (1) and N,N′-bis(ethyl-3′-benzoate)-1,3-propanediamide (2), have been synthesized and screened towards various biologically important metal ions such as Na+, Mg2+, K+, Ca2+, Al3+, Cr3+, Mn2+, Fe2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, Ag+, Cd2+, Hg2+, Ti3+, and Pb2+. The emission spectral studies of both 1 and 2 displayed 84 – 91% turn-off emission responses selectively with Fe3+ ion in aqueous buffer (MeCN/H2O, 1:4, v/v, pH = 7.4) solution. Chemosensors 1 and 2 exhibited remarkable sensing ability towards Fe3+ ion over other metal ions with limit of detection (LOD) of 4.28 and 4.33 μM, respectively. The binding stoichiometry of 1 and 2 with Fe3+ ion was studied by Benesi–Hildebrand fitting, Stern–Volmer plot and Job’s plots, revealing that both chemosensors (1 – 2) bind with Fe3+ metal ion in 1:1 stoichiometric ratio with the apparent association constant (Ka) 8.90 × 103 and 11.16 × 103 M−1, respectively. Furthermore, the interactions of chemosensors (1 – 2) with metal ion were also investigated by using density functional theory (DFT) at B3LYP hybrid functional using 6-31G and LanL2DZ basis sets.
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- vol.36, no.6, pp.647-649, 2020-06-10 (Released:2020-06-10)
- 著者
- Akira TSUJITA Asami NAGASAKA Hidehiko OKAZAKI Shin OGAWA Akinaga GOHDA Toshiro MATSUI
- 出版者
- The Japan Society for Analytical Chemistry
- 雑誌
- Analytical Sciences (ISSN:09106340)
- 巻号頁・発行日
- pp.20P042, (Released:2020-05-29)
- 被引用文献数
- 1