- 著者
- Masashi Uehara Shota Ikegami Takashi Takizawa Hiroki Oba Noriaki Yokogawa Takeshi Sasagawa Kei Ando Hiroaki Nakashima Naoki Segi Toru Funayama Fumihiko Eto Akihiro Yamaji Kota Watanabe Satoshi Nori Kazuki Takeda Takeo Furuya Sumihisa Orita Hideaki Nakajima Tomohiro Yamada Tomohiko Hasegawa Yoshinori Terashima Ryosuke Hirota Hidenori Suzuki Yasuaki Imajo Hitoshi Tonomura Munehiro Sakata Ko Hashimoto Yoshito Onoda Kenichi Kawaguchi Yohei Haruta Nobuyuki Suzuki Kenji Kato Hiroshi Uei Hirokatsu Sawada Kazuo Nakanishi Kosuke Misaki Hidetomi Terai Koji Tamai Eiki Shirasawa Gen Inoue Kenichiro Kakutani Yuji Kakiuchi Katsuhito Kiyasu Hiroyuki Tominaga Hiroto Tokumoto Yoichi Iizuka Eiji Takasawa Koji Akeda Norihiko Takegami Haruki Funao Yasushi Oshima Takashi Kaito Daisuke Sakai Toshitaka Yoshii Tetsuro Ohba Bungo Otsuki Shoji Seki Masashi Miyazaki Masayuki Ishihara Seiji Okada Yasuchika Aoki Katsumi Harimaya Hideki Murakami Ken Ishii Seiji Ohtori Shiro Imagama Satoshi Kato
- 出版者
- The Japanese Society for Spine Surgery and Related Research
- 雑誌
- Spine Surgery and Related Research (ISSN:2432261X)
- 巻号頁・発行日
- pp.2021-0183, (Released:2021-12-27)
- 被引用文献数
- 2
Background: In elderly patients with cervical spinal cord injury, comorbidities such as cardiovascular and cerebrovascular diseases are common, with frequent administration of antiplatelet/anticoagulant (APAC) drugs. Such patients may bleed easily or unexpectedly during surgery despite prior withdrawal of APAC medication. Few reports have examined the precise relationship between intraoperative blood loss and history of APAC use regarding surgery for cervical spine injury in the elderly.The presentmulticenter database survey aimed to answer the question of whether the use of APAC drugs affected the amount of intraoperative blood loss in elderly patients with cervical spinal cord trauma.Methods: The case histories of 1512 patients with cervical spine injury at 33 institutes were retrospectively reviewed. After excluding cases without spinal surgery or known blood loss volume, 797 patients were enrolled. Blood volume loss was the outcome of interest. We calculated propensity scores using the inverse probability of treatment weighting (IPTW) method. As an alternative sensitivity analysis, linear mixed model analyses were conducted as well.Results: Of the 776 patients (mean age: 75.1 ± 6.4 years) eligible for IPTW calculation, 157 (20.2%) were taking APAC medications before the injury. After weighting, mean estimated blood loss was 204 mL for non-APAC patients and 215 mL for APAC patients. APAC use in elderly patients was not significantly associated with surgical blood loss according to the IPTW method with propensity scoring or linear mixed model analyses. Thus, it appeared possible to perform surgery expecting comparable blood loss in APAC and non-APAC cases.Conclusions: This multicenter study revealed no significant increase in surgical blood loss in elderly patients with cervical trauma taking APAC drugs. Surgeons may be able to prioritize patient background, complications, and preexisting conditions over APAC use before injury when examining the surgical indications for cervical spine trauma in the elderly.
- 著者
- Kazuhide Inage Takeshi Sainoh Takayuki Fujiyoshi Otagiri Takuma Yasuchika Aoki Masahiro Inoue Yawara Eguchi Sumihisa Orita Yasuhiro Shiga Masao Koda Tsutomu Akazawa Takeo Furuya Junichi Nakamura Hiroshi Takahashi Miyako Suzuki Satoshi Maki Hideyuki Kinoshita Masaki Norimoto Tomotaka Umimura Takashi Sato Masashi Sato Masahiro Suzuki Keigo Enomoto Hiromitsu Takaoka Norichika Mizuki Takashi Hozumi Ryuto Tsuchiya Geundong Kim Tomohito Mukaihata Takahisa Hishiya Seiji Ohtori
- 出版者
- The Japanese Society for Spine Surgery and Related Research
- 雑誌
- Spine Surgery and Related Research (ISSN:2432261X)
- 巻号頁・発行日
- pp.2020-0042, (Released:2020-07-10)
- 被引用文献数
- 3
Introduction: Mirogabalin should be equivalent to pregabalin, but with fewer incidences of adverse drug reactions (ADRs). To verify these benefits in actual clinical trials, our study investigated the frequency of ADRs and mirogabalin' s analgesic effects during treatment of peripheral neuropathic pain.Methods: This study included 74 patients with lower limb pain. We surveyed patient reports of ADRs during the follow-up period as the primary endpoint and examined the visual analog scale (VAS) reported for lower limb pain as the secondary endpoint (before administration, and two and four weeks after administration).Results: The occurrence of ADR was 27.0%, like the frequency of ADRs in the clinical trials for other disorders. However, the discontinuation rate of administration was 10.8%, which was significantly lower than the frequency of ADR occurrences. When the analgesic effect was assessed, a significant decrease in the temporal change of VAS for lower limb pain was observed before administration, and two and four weeks after administration.Conclusions: In this study, the occurrence of ADRs reported by the patients was like the frequency of ADRs reported in the clinical trials for other disorders. When assessing the analgesic effect, the temporal change of VAS for lower limb pain was found to decrease significantly before administration, and two and four weeks after administration.
- 著者
- Yasuhiro Shiga Sumihisa Orita Kazuhide Inage Jun Sato Kazuki Fujimoto Hirohito Kanamoto Koki Abe Go Kubota Kazuyo Yamauchi Yawara Eguchi Masahiro Inoue Hideyuki Kinoshita Yasuchika Aoki Junichi Nakamura Yusuke Matsuura Richard Hynes Takeo Furuya Masao Koda Kazuhisa Takahashi Seiji Ohtori
- 出版者
- The Japanese Society for Spine Surgery and Related Research
- 雑誌
- Spine Surgery and Related Research (ISSN:2432261X)
- 巻号頁・発行日
- vol.1, no.4, pp.197-202, 2017-10-20 (Released:2017-11-27)
- 参考文献数
- 24
- 被引用文献数
- 22 25
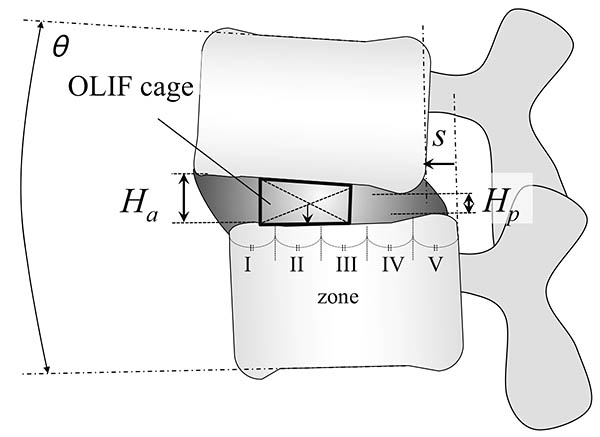
Introduction: Oblique lateral interbody fusion (OLIF) can achieve recovery of lumbar lordosis (LL) in minimally invasive manner. The current study aimed to evaluate the location of lateral intervertebral cages during OLIF in terms of LL correction.Methods: The subjects were patients who underwent OLIF for lumbar degenerative diseases, including lumbar spinal stenosis, spondylolisthesis, and discogenic low back pain. Their clinical outcome was evaluated using visual analogue scale on lower back pain (LBP), leg pain and numbness. The following parameters were retrospectively evaluated on plain radiographic images and computed tomography scans before and at 1 year after OLIF: the intervertebral height, vertebral translation, and sagittal angle. The cage position was defined by equally dividing the caudal endplate into five zones (I to V), and its association with segmental lordosis restoration was analyzed. Subjects were also evaluated for a postoperative endplate injury.Results: Eighty patients (121 fused levels) with lumbar degeneration who underwent OLIF were included. There were no significant specific distribution in preoperative disc pathology such as disc angle, height, and translation. After OLIF, sagittal alignment was improved with an average correction angle of 3.8º at the instrumented segments in a level-independent fashion. All cases showed significant improvement in clinical outcomes, and had improvement in the radiological parameters (P<0.05). A detailed analysis of the cage position showed that the most significant sagittal correction and the most postoperative endplate injuries occurred in the farthest anterior zone (I). Cages with a 12-mm height were associated with more endplate injuries compared with shorter cages (8 or 10 mm).Conclusions: OLIF improves sagittal alignment with an average correction angle of 3.8º at the instrumented segments. We suggest that the optimal cage position for better lordosis correction and the fewest endplate injuries is zone II with a cage height of up to 10 mm.
- 著者
- Koji Tamai Hidetomi Terai Akinobu Suzuki Hiroaki Nakamura Masaomi Yamashita Yawara Eguchi Shiro Imagama Kei Ando Kazuyoshi Kobayashi Morio Matsumoto Ken Ishii Tomohiro Hikata Shoji Seki Masaaki Aramomi Tetsuhiro Ishikawa Atsushi Kimura Hirokazu Inoue Gen Inoue Masayuki Miyagi Wataru Saito Kei Yamada Michio Hongo Kenji Endo Hidekazu Suzuki Atsushi Nakano Kazuyuki Watanabe Junichi Ohya Hirotaka Chikuda Yasuchika Aoki Masayuki Shimizu Toshimasa Futatsugi Keijiro Mukaiyama Masaichi Hasegawa Katsuhito Kiyasu Haku Iizuka Kotaro Nishida Kenichiro Kakutani Hideaki Nakajima Hideki Murakami Satoru Demura Satoshi Kato Katsuhito Yoshioka Takashi Namikawa Kei Watanabe Kazuyoshi Nakanishi Yukihiro Nakagawa Mitsunori Yoshimoto Hiroyasu Fujiwara Norihiro Nishida Masataka Sakane Masashi Yamazaki Takashi Kaito Takeo Furuya Sumihisa Orita Seiji Ohtori
- 出版者
- The Japanese Society for Spine Surgery and Related Research
- 雑誌
- Spine Surgery and Related Research (ISSN:2432261X)
- 巻号頁・発行日
- vol.1, no.4, pp.179-184, 2017-10-20 (Released:2017-11-27)
- 参考文献数
- 26
- 被引用文献数
- 3 3
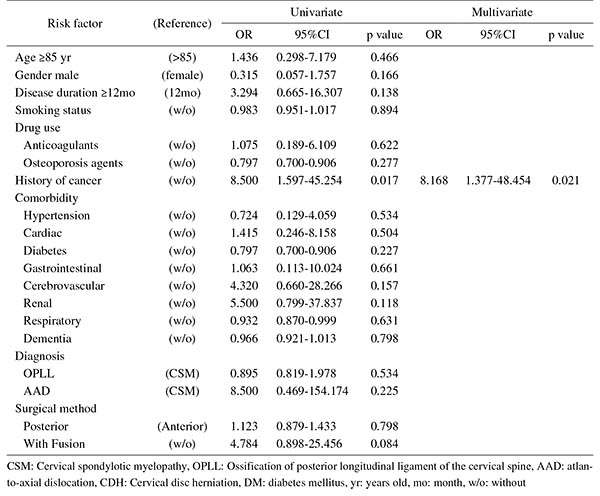
Introduction: With an aging population, the proportion of patients aged ≥80 years requiring cervical surgery is increasing. Surgeons are concerned with the high incidence of complications in this population, because "age" itself has been reported as a strong risk factor for complications. However, it is still unknown which factors represent higher risk among these elderly patients. Therefore, this study was conducted to identify the risk factors related to surgical complications specific to elderly patients by analyzing the registry data of patients aged ≥80 years who underwent cervical surgery.Methods: We retrospectively studied multicenter collected registry data using multivariate analysis. Sixty-six patients aged ≥80 years who underwent cervical surgery and were followed up for more than one year were included in this study. Preoperative patient demographic data, including comorbidities and postoperative complications, were collected from multicenter registry data. Complications were considered as major if they required invasive intervention, caused prolonged morbidity, or resulted in prolongation of hospital stay. Logistic regression analysis was performed to analyze the risk factors for complications. A p-value of <0.05 was considered as statistically significant.Results: The total number of patients with complications was 21 (31.8%), with seven major (10.6%) and 14 minor (21.2%) complications. Multivariate logistic regression analysis, after adjusting for age, revealed two significant risk factors: preoperative cerebrovascular disorders (OR, 6.337; p=0.043) for overall complications and cancer history (OR, 8.168; p=0.021) for major complications. Age, presence of diabetes mellitus, and diagnosis were not significant predictive factors for complications in this study.Conclusions: Preoperative cerebrovascular disorders and cancer history were risk factors for complications after cervical surgery in patients over 80 years old. Surgeons should pay attention to these specific risk factors before performing cervical surgery in elderly patients.
- 著者
- Mitsuru Yagi Nobuyuki Fujita Tomohiko Hasegawa Gen Inoue Yoshihisa Kotani Seiji Ohtori Sumihisa Orita Yasushi Oshima Daisuke Sakai Toshinori Sakai Hiroshi Taneichi Daisuke Togawa Kazuo Nakanishi Hiroaki Nakashima Toshitaka Yoshii Masaya Nakamura Motoki Iwasaki Masahiko Watanabe Hirotaka Haro Tokumi Kanemura Naobumi Hosogane New Technology Assessment Committee of The Japanese Society for Spine Surgery and Related Research
- 出版者
- The Japanese Society for Spine Surgery and Related Research
- 雑誌
- Spine Surgery and Related Research (ISSN:2432261X)
- 巻号頁・発行日
- pp.2022-0194, (Released:2022-12-12)
- 被引用文献数
- 1
IntroductionLateral lumbar interbody fusion (LLIF) has been introduced in Japan in 2013. Despite the effectiveness of this procedure, several considerable complications have been reported. This study reported the results of a nationwide survey performed by the Japanese Society for Spine Surgery and Related Research (JSSR) on the complications associated with LLIF performed in Japan.MethodsJSSR members conducted a web-based survey following LLIF between 2015 and 2020. Any complications meeting the following criteria were included: (1) major vessel, (2) urinary tract, (3) renal, (4) visceral organ, (5) lung, (6) vertebral, (7) nerve, and (8) anterior longitudinal ligament injury; (9) weakness of psoas; (10) motor and (11) sensory deficit; (12) surgical site infection; and (13) other complications. The complications were analyzed in all LLIF patients, and the differences in incidence and type of complications between the transpsoas (TP) and prepsoas (PP) approaches were compared.ResultsAmong the 13,245 LLIF patients (TP 6,198 patients [47%] and PP 7,047 patients [53%]), 389 complications occurred in 366 (2.76%) patients. The most common complication was sensory deficit (0.5%), followed by motor deficit (0.43%) and weakness of psoas muscle (0.22%). Among the patient cohort, 100 patients (0.74%) required revision surgery during the survey period. Almost half of the complications developed in patients with spinal deformity (183 patients [47.0%]). Four patients (0.03%) died from complications. Statistically more frequent complications occurred in the TP approach than in the PP approach (TP vs. PP, 220 patients [3.55%] vs. 169 patients [2.40%]; p < 0.001).ConclusionsThe overall complication rate was 2.76%, and 0.74% of the patients required revision surgery because of complications. Four patients died from complications. LLIF may be beneficial for degenerative lumbar conditions with acceptable complications; however, the indication for spinal deformity should be carefully determined by the experience of the surgeon and the extent of the deformity.